CORCEPT THERAPEUTICS (CORT)·Q4 2025 Earnings Summary
Corcept Misses Q4, But 2026 Guidance and July PDUFA Loom Large
February 24, 2026 · by Fintool AI Agent
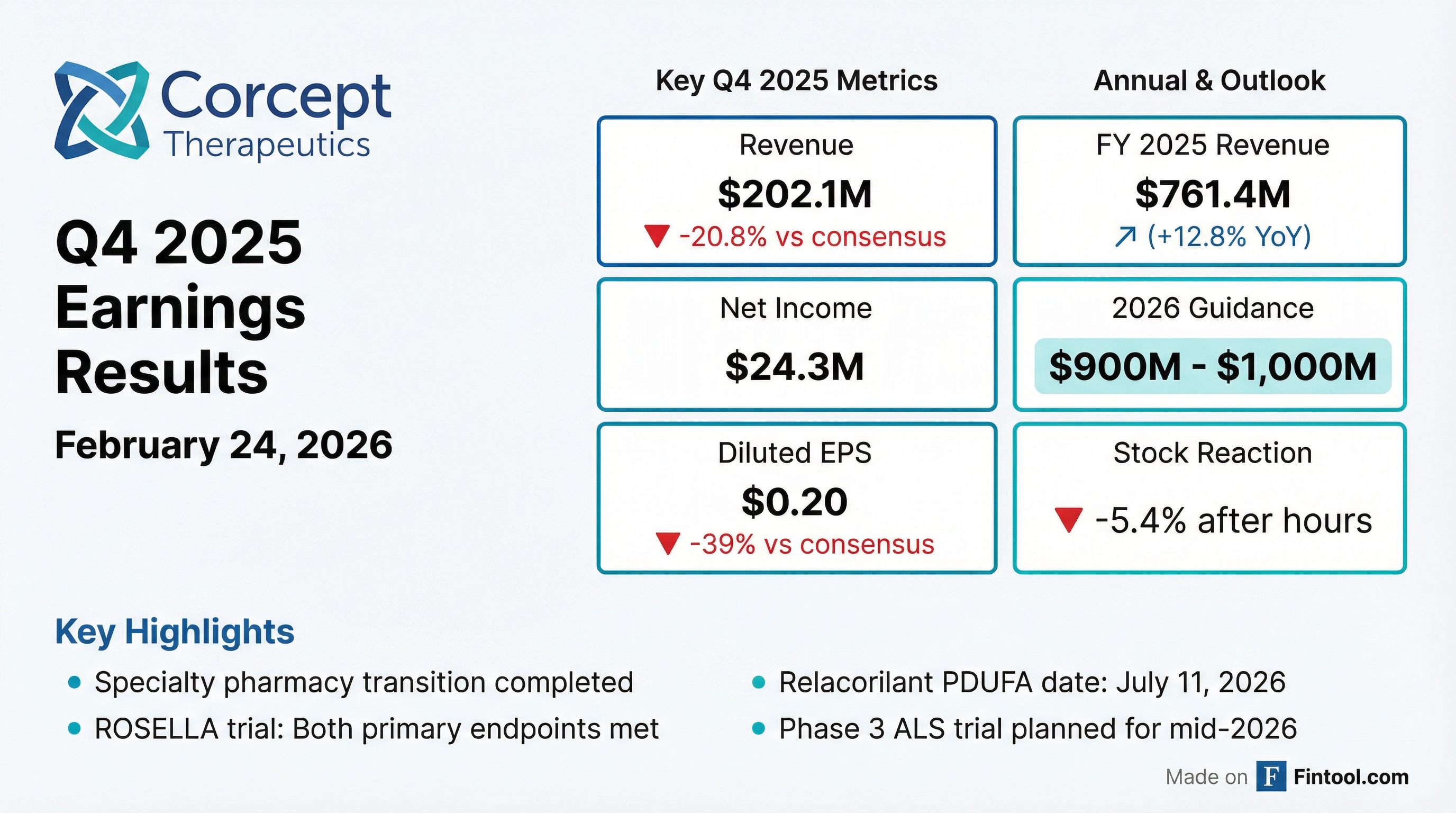
Corcept Therapeutics (NASDAQ: CORT) reported Q4 2025 results that missed Wall Street expectations by a wide margin, with revenue of $202.1 million and diluted EPS of $0.20 falling well short of consensus. Management attributed the shortfall to specialty pharmacy transition disruptions but struck an optimistic tone on 2026, guiding to $900M-$1B in revenue and highlighting a July PDUFA date for relacorilant in ovarian cancer.
The stock traded down ~5.4% in after-hours following the release, reflecting disappointment in Q4 execution despite the forward-looking catalysts.
Did Corcept Beat Earnings?
No. Corcept missed on both revenue and EPS:
*Values retrieved from S&P Global
The miss was driven by operational disruptions during Q4 as Corcept transitioned to a new specialty pharmacy vendor. CEO Joseph Belanoff acknowledged the company "should have achieved higher growth but were not able to fully meet demand because of capacity constraints at our previous specialty pharmacy vendor."
Despite the Q4 stumble, tablets sold grew 37% YoY, and the company saw record new prescriptions and prescribers for the full year.
How Did Full Year 2025 Compare to 2024?
Revenue grew despite the Q4 challenges, but profitability declined sharply. The company spent $245.9 million on share repurchases during 2025, contributing to the decline in cash.
What Did Management Guide for 2026?
Corcept provided FY 2026 revenue guidance of $900M to $1,000M, implying 18-31% growth over 2025.
Key assumptions behind the guidance:
- Specialty pharmacy transition complete — Management stated the transition is "now fully complete" and February is tracking toward a record month for new patient starts
- Cushing's syndrome demand recovery — Growing physician recognition of hypercortisolism prevalence
- Potential relacorilant approvals — PDUFA date of July 11, 2026 for platinum-resistant ovarian cancer
The guidance notably does not include revenue from relacorilant in new indications, suggesting upside optionality if approvals materialize.
How Did the Stock React?
The stock has been under significant pressure, trading 69% below its 52-week high. The after-hours decline suggests investors were disappointed by Q4 execution, though the guidance and clinical catalysts may provide support.
What Changed From Last Quarter?
Positive developments:
- Specialty pharmacy transition completed (was ongoing in Q3)
- ROSELLA trial met both primary endpoints (progression-free and overall survival)
- GRACE Phase 3 results published in The Lancet Diabetes & Endocrinology
- MOMENTUM trial enrollment completed (1,000 patients with resistant hypertension)
Areas of concern:
- Q4 revenue significantly impacted by operational issues
- Net income declined 21% QoQ ($24.3M vs $30.7M in Q4 2024)
- Relacorilant for Cushing's received Complete Response Letter — still working with FDA
What Are the Key Upcoming Catalysts?
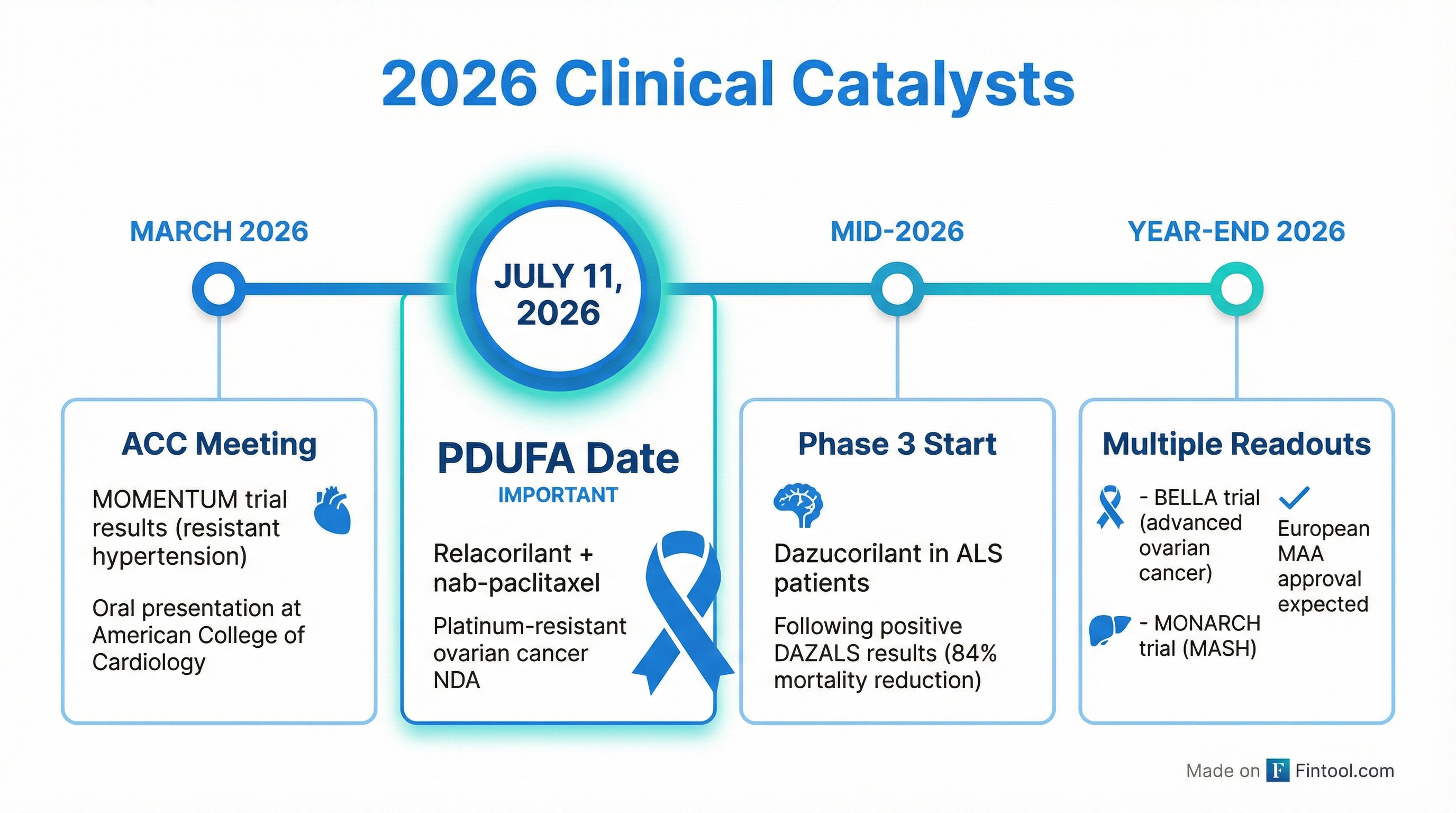
The July 11 PDUFA date for relacorilant in platinum-resistant ovarian cancer is the most significant near-term catalyst. The ROSELLA trial demonstrated a 35% reduction in risk of death without biomarker selection or increased safety burden.
What Did Management Say About ALS?
The DAZALS trial for dazucorilant in ALS patients showed remarkable results: 84% reduction in risk of death during the first year compared to placebo (hazard ratio: 0.16, p-value: 0.0009).
Management is conducting a dose titration study to improve gastrointestinal tolerability before initiating a Phase 3 trial by mid-2026.
This represents a potential new therapeutic area for Corcept beyond Cushing's syndrome and oncology.
Historical Beat/Miss Track Record
Looking at Corcept's recent earnings history:
Corcept has shown volatility in meeting revenue expectations, particularly in Q4 quarters (Q4 2024 and Q4 2025 both missed significantly). EPS has been more consistently above expectations outside of Q4 periods.
Key Risks and Concerns
-
FDA pathway uncertainty — Relacorilant for Cushing's syndrome received a Complete Response Letter; timeline to approval remains unclear
-
Execution risk — Q4 demonstrated operational challenges can materially impact results
-
Concentration risk — Cushing's syndrome business is the primary revenue driver; relacorilant approvals needed to diversify
-
Cash consumption — Cash declined $71M YoY while company continues heavy share repurchases
Bottom Line
Q4 2025 was a clear operational miss for Corcept, but the forward story remains intact. The specialty pharmacy transition is complete, February patient starts are tracking to records, and the 2026 catalyst calendar is packed. The July 11 PDUFA date for relacorilant in ovarian cancer is the next major inflection point—approval would validate the cortisol modulation platform beyond Cushing's and open a significant new market.
At $3.8B market cap and trading 69% off highs, the stock prices in considerable skepticism. Bulls will focus on the $900M-$1B guidance and multiple shots on goal; bears will point to the Complete Response Letter and Q4 execution failure as reasons for caution.
Earnings call scheduled for February 24, 2026 at 5:00 PM ET. View company profile | View documents